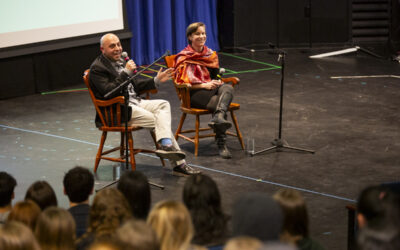On the screen in the Performing Arts Center, a pair of photographs, taken a day apart in 2012, documented the moment that changed everything for the Ganot family. Annie and Ilan Ganot P’26 had been pursuing careers in investment banking in London with their young children Eliya ’26 and Eytani. An image taken on October 14 showed the siblings mugging for the camera, dressed for a Halloween party. The picture from October 15 was just of Eytani, in a hospital gown—it was the day they learned he had muscular dystrophy and wasn’t expected to live beyond 30. A few weeks later, the Ganots were hatching plans to move to Boston to start a drug development company that will try to help patients with this condition.
As they shared in their presentation to CA students on November 29, 2023, though they had no prior experience in the health care industry, the Ganots knew they could help accelerate the pace of exploratory treatments for Duchenne muscular dystrophy—the most common type of muscular dystrophy, which Eytani was diagnosed with. This progressive, life-limiting muscle-wasting disease is untreatable and rare, affecting between 1 in 3,500 to 1 in 5,000 newborns, almost exclusively those born with XY chromosomes. But rarity is relative: Roughly 10,000 to 15,000 children in the U.S. are living with Duchenne today.
With considerable warmth and candor, the Ganots helped the CA community understand Duchenne’s genetic origin and the course of the disease. A mutation in a single gene prevents the production of a protein, dystrophin, which is required for normal muscle function. Duchenne is often passed from mother to child (those with XX chromosomes are usually carriers, who may not be aware of the possibility), but it can also result from random spontaneous genetic mutation during any pregnancy. By the time children with Duchenne reach their early teens, Annie explained, they’re no longer able to walk; by their early 20s, they lose upper body function. As she said, Eytani will “lose his ability to feed himself and to hug his mother.”
The Ganots gave students insight into the business acumen that allowed them to build a biotechnology company that has garnered significant funding from investors to develop promising treatments for Duchenne. They shared how they leveraged their expertise, networks, and unrelenting drive as parents and professionals to push boundaries in science and technology to improve the lives of all individuals living with this condition.
When they founded Solid Biosciences, a Boston-based biotechnology company focused on developing treatments for Duchenne, in 2013, the Ganots raised initial money which enabled them to hire Solid’s first scientist with Duchenne research expertise and rent shared office space at the Cambridge Innovation Center. After initially focusing on many treatment pathways, Ilan said, they quickly narrowed their focus to gene therapy, based on “remarkable outcomes” from early preclinical work.
By 2018, they had several dozen employees and began a clinical trial in patients with Duchenne. Ilan spoke about his approach to limiting his involvement to the business side of the company: For the medical research itself, “you hire really smart people and then get out of their way.”


That year, they also took the company public. “It’s a lot more fun when it’s private,” Ilan quipped, “but you have access to more capital when it’s public.” And that funding is indispensable—Ilan called the treatment trials “very expensive experiments.”
Clinical trials assess the efficacy of a new drug treatment while considering its safety profile, he explained. While there were signs of benefit in treated patients in the study, there were also safety concerns that the company managed over those critical years. In 2022, after the company had grown further and then been scaled back, Solid Biosciences merged with another gene therapy company, and Ilan, needing a break, stepped down as CEO.
The couple remains highly involved in what Annie called the “Duchenne community” of families whose lives have been restructured around caring for their children. Annie highlighted the unwavering support network they have built and the many fundraising events that also gather and support this community. She directed students who wish to learn more to the Jett Foundation and Parent Project Muscular Dystrophy.
As a patient advocate at Solid Biosciences, Annie works directly with patients and their families. She helps those who have just learned about Duchenne navigate the road ahead as a fellow parent who intimately understands how their lives have suddenly split, as she said, into “pre-diagnosis and post-diagnosis.” She helps reduce their burdens and anxiety, creates tools to enable children to envision what participating in clinical trials will be like, and ensures that patients’ perspectives are always centered in the company’s work.
“It’s a privilege to work every day in a job to improve the human condition,” Annie said. What is cooler than that?”
The Ganots’ assembly was particularly timely. Just weeks earlier, another, next-generation Solid Biosciences gene therapy candidate had received FDA permission to initiate clinical trials. And the same day the Ganots spoke at CA, the United Nations officially voted to designate September 7 as World Duchenne Awareness Day.
“For us as a family, this has been an opportunity to truly try to make a difference in our son’s life and the lives of other boys with Duchenne,” Annie said. “We know what matters, and that gift of perspective has been life-changing for us.”